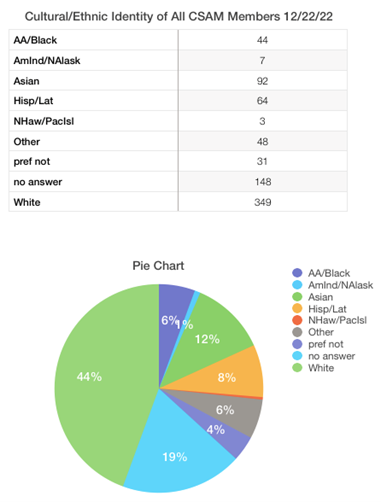
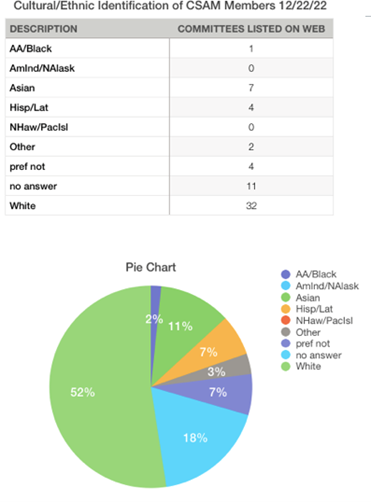
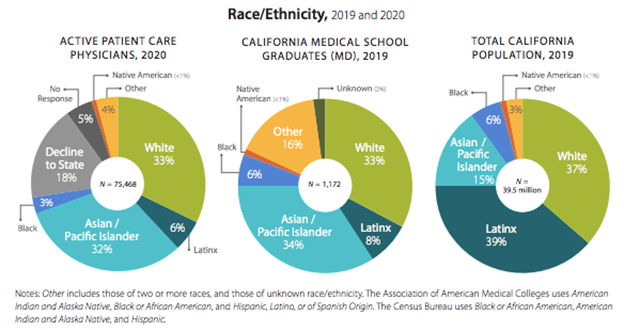
Dear CSAM Members,
I am pleased to announce the appointment of Cheryl Marcell as the new Executive Director of CSAM, effective June 15, 2024. Cheryl joins us through a contract with McDonald Association Management in Sacramento, CA, bringing over two decades of distinguished experience and a diverse skill set acquired through her leadership roles with nonprofit organizations in Sacramento and Canada.
Cheryl’s impressive background includes expertise in advocacy at both state and federal levels, membership services, cultivating strategic relationships, revenue generation, and managing conferences and events. She is a seasoned communicator, adept at representing organizations to the public, government, and the media. Cheryl’s most recent role involved serving a large nonprofit that provides crucial services to victims of human trafficking and domestic violence.
McDonald Association Management boasts a skilled team with a proven track record of over two decades in association management. Their expertise spans project management, member services, financial management, event planning, grant development, and government affairs.
The recommendation of McDonald was the culmination of an extensive search process that commenced in February. The search committee, led by Chair Dr. Dana Harris and comprising committee members Dr. Jean Marsters, Dr. Karen Miotto, Dr. Mario San Bartolome, Dr. Mason Turner, Dr. Neal Mehra, and myself, carried out the search process. Kerry Parker, CSAM’s Executive Director 2000-2020, served as our management search consultant.
As part of this new partnership with McDonald, CSAM’s new office and mailing address will be 1540 River Park Drive, Suite 211, Sacramento, CA, 95815-4608, while the primary email address (csam@csam-asam.org) will remain unchanged.
In addition to partnering with McDonald, CSAM has also engaged ConferenceDirect, a reputable nationwide conference management company, to provide support for the 2024 annual conference. Joanna Ward, serving as the conference manager, is collaborating closely with the conference planning committee on the State of the Art Addiction Medicine Conference coming up August 21-24 at the Hyatt Regency San Francisco.
I am eagerly anticipating the conference and the opportunity to introduce our members to Cheryl and our new management team. I look forward to the pleasure of seeing many of you there.
Warm regards,
Sharone Abramowitz, MD, DFASAM
President, California Society of Addiction Medicine